 The third IE, however, is over five times the previous one. Again, this is due to the effective charge at the nucleus. AP Chem > Unit 1 1.7 Periodic Trends 8 min read decemJeremy Kiggundu Dalia Savy Share A cool thing about the periodic table is that it is organized to demonstrate different trends and properties of elements that can be explained by the pattern of electron configurations and the presence of electron-filled orbitals. This direction increases the size of the atom. The second IE is twice the first, which is not a surprise: the first IE involves removing an electron from a neutral atom, while the second one involves removing an electron from a positive ion. The other trend of atomic radius or atom size occurs as you move vertically down an element group. The closer the electrons are to the nucleus, the smaller the distance. 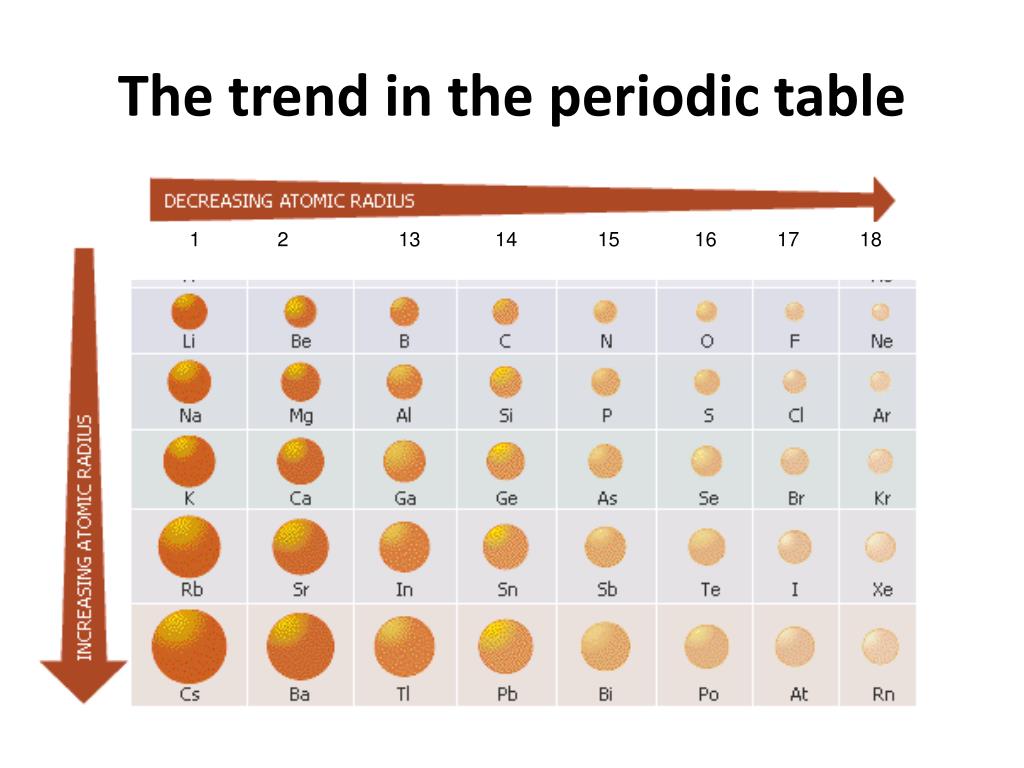
It increases as you move from left to right across the periodic table. Electrons not in the valence shell shield the valence electrons from the feeling the full effect of the nucleus. This means that there is a higher nuclear charge which increases the pull the nucleus has on the electrons. The charge of the nucleus felt by the valence electrons. Although there are some reversals in the trend (e.g., see Po in the bottom row), atoms generally get smaller as you go across the periodic table and larger as you go down any one column. Study with Quizlet and memorize flashcards containing terms like Zeff (effective nuclear charge) equation, Effective Nuclear Charge, Effective nuclear charge trend and explanation (Coulombs Law) across and more. The first ionization energy of K is less than that of Na. These molecules have a trigonal planar structure. Boron commonly forms molecules of the type BX3. (a) (b) (c) (d) Atomic size decreases from Na to Cl in the periodic table. As you go right, the atomic numbers increase. 1: Atomic Radii Trends on the Periodic Table. Account for each of the following observations in terms of atomic theory and/or quantum theory. (b) The metallic atomic radius, r met, is half the distance between the nuclei of two adjacent atoms in a pure solid metal, such as aluminum.(g) + e^− \nonumber \] Going from left to right on the periodic table, the atomic radii get smaller.  
The radius increases as you move down a group (column) and decreases as you move from left to right across a period (row). (a) The covalent atomic radius, r cov, is half the distance between the nuclei of two like atoms joined by a covalent bond in the same molecule, such as Cl 2. Both atomic radius and ionic radius follow a trend on the periodic table. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
